Fragrances are used in countless consumer products, from perfume to soap, and even sunscreen. The chemical details behind these fragrances dictate how the fragrance will smell and how long the scent will last. Luckily, we were able to interview Mr. Herman, a cosmetic chemist, who was able to elaborate on the complexities of fragrance chemistry.

During the interview, we hoped to find out:
What makes up a fragrance?
How does the chemistry influence a scent?
What miniscule chemical interactions are taking place?
BCAH1: What are the main components of a fragrance?
Mr. Herman: In the fragrance world as a whole, the biggest class of fragrances is made up of alcohols and the second biggest class is made up of aldehydes and ketones. RIFM (The Research Institute for Fragrance Materials) conducts a census every two years to gain information on what chemicals fragrance companies buy. A fragrance will most likely be a mix of natural and synthetic products. There are certainly more synthetic products which you can get creative with. The quality control and consistency of synthetics are great. However, natural products can be hard to replicate well. Sometimes it is easier to make a synthetic replicating a natural ingredient like citrus oil.
Although virtually clear to the human eye, perfumes are quite complex. Many components of a perfume are found in nature, like aldehydes and ketones which are oxygen- containing hydrocarbons. These molecules have low molecular weights and have solvent characteristics such as acting as a medium capable of dissolving other substances.
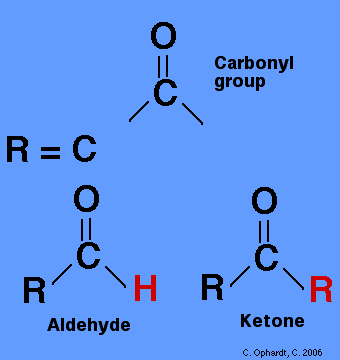
Figure 1: This is a visual representation of aldehyde and ketone. Both are organic compounds. Each structure contains a carbon double bonded to an oxygen (carbonyl group). The carbon has two remaining bonds. If one element bonded to the carbonyl is a hydrogen, the substance is an aldehyde. If not, the substance is a ketone. These functional groups contain polar bonds and can thus dissolve other polar compounds.

Figure 2: This image contains natural aldehydes and ketones. Synthetic products are produced based on the these natural molecules found throughout the world.
Larger aldehydes like cinnamaldehyde tend to have a pleasant smell, whereas the smaller aldehydes have pungent, sharp smells. Aldehydes are derived from alcohols by dehydrogenation. Dehydrogenation is the removal of a hydrogen from a chemical compound.
BCAH1: What effect does light have on fragrance bonds?
Mr. Herman: It has an effect on stability. Alot of things affect fragrance stability. For example, colors affect stability. UV will trigger changes, but beyond that is oxidation. Oxidation is probably the worst thing that can happen to a fragrance. Even when you put a concentrated fragrance into alcohol or base, they can still change. A phenol component is very reactive, it changes color easily because it is easy to take the hydrogen off, leaving a free radical which reacts with all kinds of stuff. This phenol component is fine in the dark, but in the light, the hydrogen gets knocked off and makes the compound very reactive. Chelating agents (EDTA disodium salt), benzophenones (UV absorbers), and antioxidants are added to fragrances to stabilize them. Oxidation is the worst, followed by heat and sunlight because they catalyze reactions. pH is a whole other topic. They relate to esters, which break apart in extreme pH levels. All these factors are important to consider in making a fragrance stable.
Oxidation/Reduction Reactions
Oxidation/Reduction Reactions are chemical reactions where the oxidation state of atoms is changed. It involved the transfer of electrons. Aldehydes are strong reducing agents because of the presence of the hydrogen atom. On the other hand, ketones are resistant to being oxidized. Only very strong oxidizing reagents have the power to oxidize ketones like potassium manganate (VII) solution by destructively breaking the carbon to carbon bond. Aldehydes (RCHO) are mostly oxidized into carboxylic acids (RCO2H).
The following reactions display the half reaction of oxidation for:
An aldehyde under acidic conditions:
RCHO + H2O → RCOOH + 2 H++ 2 e-
An aldehyde under basic conditions:
RCHO + 3OH- → RCOO- + 2H2O +2 e –
BCAH1:Discuss how size of molecules affects fragrance.
Mr. Herman: Fragrance molecules must be relatively small. The biggest molecule are musks, with the biggest molecular weight of about 350. These are humongous! Most fragrance molecules have a molecular weight of about 120 to 180. Those molecules have a group coming off of it known as an osmophore. Osmophores are floral fragrance glands which are responsible for making things “smell”.
Aldehydes with larger molecular weights tend to have pleasant, flowery scents.
Different Senses in One Perfume:
There are 3 different notes which unfold after the perfume is sprayed: top notes, heart notes, base note
1. Top Notes
Top notes are smelt immediately after a perfume bottle is sprayed to up to 15 minutes . Since the top notes reach your nose instantly, these molecules must be small and light enough to evaporate fast
2. Heart Notes
Heart notes may appear within 2 minutes to an hour after application. These compounds emerge in the middle of the perfume’s dispersion process
3. Base Notes
Base notes are the most heavy of molecules that tend to “stick” to your skin. These molecules can smelt for hours after first application.

BCAH1: What are the intermolecular forces at play within a fragrance?
Mr. Herman: The simplest way of looking at the polarity of molecules is that they are either water soluble or oil soluble, and that they are polar or nonpolar. Most fragrances can mix together. There are some limitations however, starting with the number of crystals you have. If you have too many crystals, they will fall out of solution. They can make up a maximum of 35% of the fragrance. Solvents are added to fragrances, and they affect the polarity of the solution. Usually, DPG (dipropylene glycol — colorless odorless solvent) is added. Nonpolar things, such as diethyl phthalate, must also be added to keep everything in solution. Diethyl phthalate, however, has a bad reputation as an endocrine disrupter. If you want to add a fragrance to a shampoo, you need a polar substance, where if you are adding fragrance to a lipstick, you need a nonpolar substance. Perfumes and fragrances can vary from polar and nonpolar. Believe it or not, water soluble fragrances aren’t actually water soluble. You just add surfactants!* A fragrance can either be polar with something like DPG, nonpolar, or water soluble with a surfactant. We usually use nonionic surfactants. A surfactant is a surface-active agent, similar to emulsifiers. Basically you are dealing with surface tension. A drop of water is shaped like a bead. The surface is not inert, the attractive forces in the molecule are big enough to overcome gravity. If you add a detergent to the drop, it spreads out because you effect the surface tension. In a perfume, you want to reduce the surface tension. You add some mechanical energy to the solution, and then you get the emulsion
*Surfactants are responsible for reducing the surface tension of certain components in fragrances. By weakening the strong intermolecular forces that induce surface tension, it is easier to mix different substances.
In conclusion, chemistry is able to explain the function of cosmetics, especially fragrances. Thanks to Mr. Herman, we were able to learn more about the composition of fragrances and perfumes. Fragrances are composed of oils, solvents, and aroma compounds. These compounds, polar and nonpolar, mix together through emulsion, allowing consumers to enjoy many desired properties.